BOPA SACT Verification HUB (including the passport)

Welcome to the BOPA SACT Verification Hub. On this page you will find quick links to all items needed to get yourself or your trainees verified to clinically check SACT prescriptions in line with the BOPA Standards.
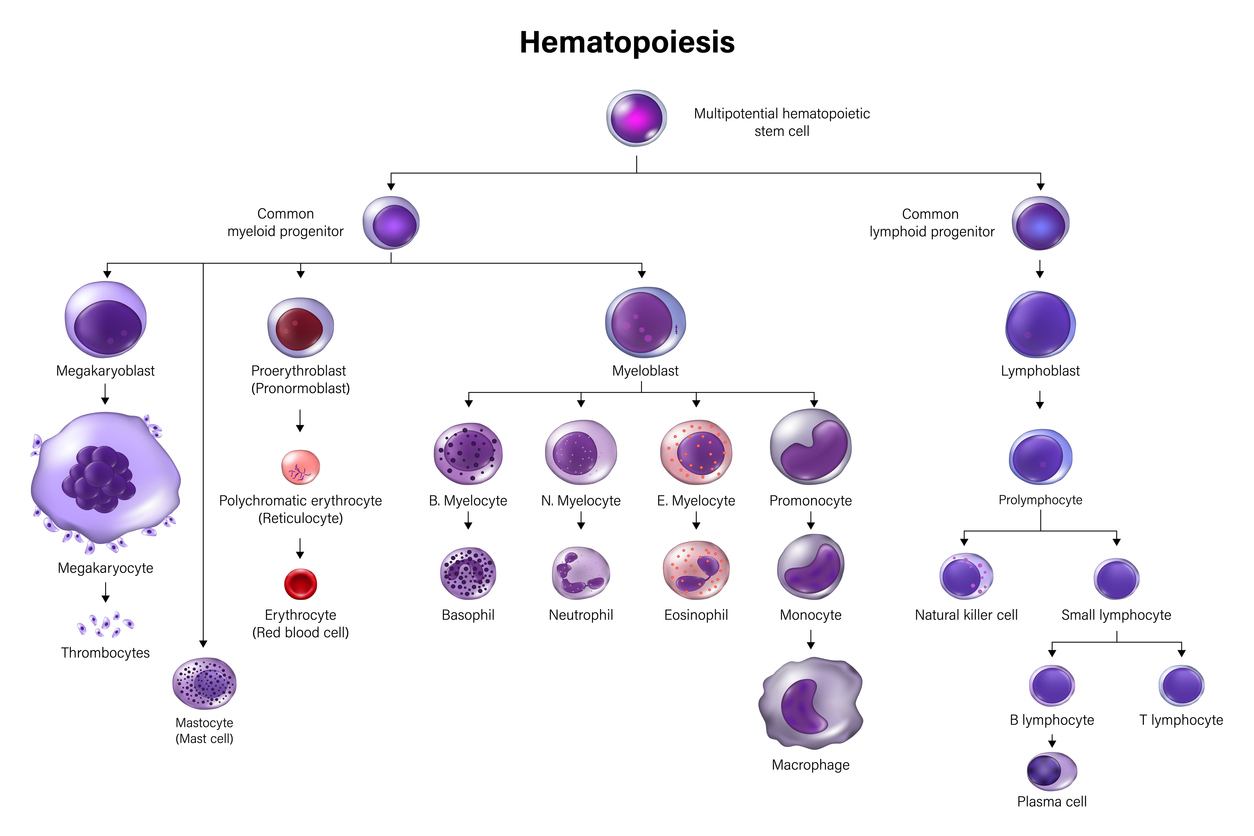
The BOPA SACT Verification Passport currently supports verification for:
- Adult Solid Tumour
- Adult Haematology
- Paediatric Solid Tumour
- Paediatric Haematology
- HSCT: Adult Autologous Transplantation
- HSCT: Adult Allogeneic Transplantation
- HSCT: Adult CAR-T
You need to be a FREE ASSOCIATE of BOPA to access this content. To sign up click here: https://www.bopa.org.uk/join-bopa/
It is recommended that you read the following before starting on your BOPA Digital SACT Verification Passport journey:
- The BOPA Education & Training Standards for Registered Pharmacy Professionals working in Cancer Services – Explains levels of training within oncology pharmacy
- The BOPA Standards for the Pharmacy Verification of Prescriptions for Cancer Medicines – The minimum standards required to check a SACT prescription (with a checklist)
- The BOPA Digital SACT Verification Passport – Set up and user guide– How to get started with the passport
If you have any questions please contact [email protected]
Please do not set up any dummy accounts in your organisation. All documents and videos are on the website in this hub to review. The only thing you cannot access are the MCQs and actual mock prescriptions (but the summary of titles are in the hub).
With thanks to the many pharmacists and pharmacy technicians who made this passport possible.